CHICAGO/LONDON – When French drugmaker Sanofi published the results of clinical trials of children given its dengue vaccine two years ago, the overall findings were that it protected against the world’s biggest and fastest growing mosquito-borne disease.
But the trial also showed that in the third year after receiving the Dengvaxia inoculation, younger children were more likely to end up in hospital with a severe case of dengue than those who didn’t get the vaccine.
The study’s authors cited two main possibilities: the children had immature immune systems that made the vaccine less protective, or the vaccine itself made them more susceptible to severe disease if they had never had dengue and later became infected.
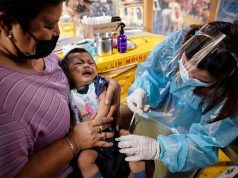
More than two years later, it turns out the latter was the primary factor – a revelation at the end of last month that has triggered alarm among hundreds of thousands of anxious parents in the Philippines, where the vaccine has been given to over 830,000 children.
It has also torpedoed initial expectations by analysts and the company that Dengvaxia, the first dengue vaccine to be developed, might become a $1 billion-a-year blockbuster product. Sanofi says it will take a charge of around 100 million euros ($118 million) for the fourth quarter.
Initially, to address the issue seen in younger children, Sanofi had recommended that the vaccine only be given to those aged 9 and older in areas where the disease was widespread. The World Health Organization analyzed Sanofi’s data and came to the same conclusion. It made a conditional recommendation to use the vaccine.
But after a new analysis of data from the trials, Sanofi confirmed last month the vaccine could increase the risk of severe dengue in some cases in people who had not been previously exposed to the disease. The WHO has now backed a decision by the Philippines government to suspend a mass immunization program and said it has begun reviewing safety data.
NO BLOOD SAMPLES COLLECTED
In Sanofi’s large-scale trials, blood samples were not collected from all the children before they were vaccinated, which would have identified prior exposure to the disease by showing the presence of antibodies.
“The development process around the first dengue vaccine led to a degree of momentum and judgments being made that we should learn lessons from,” Neil Ferguson, a professor at Imperial College London and an unpaid adviser to both Sanofi and the WHO, told Reuters.
The case raises questions whether Sanofi and the WHO, in their pursuit of a new weapon to fight a deadly disease, should have foreseen the risk. Their decisions on how the vaccine was rolled out could set back efforts to combat dengue by a generation, some disease experts say.
REJECTED SUGGESTIONS
Sanofi has rejected suggestions it ignored any risks or took any short-cuts. However, it has acknowledged that clinical tests of the vaccine did not fully investigate whether a previous dengue infection could influence the outcome.
”When you are the first in class, we’re the ones having to develop and understand the science as we go,“ said Dr. Su-Peing Ng, the global medical head of Sanofi Pasteur, the vaccines division of the drugmaker. ”We don’t have another vaccine to follow the lead on.
“The findings we have today, we would have loved to have (earlier),” she told Reuters. “Now that we have these findings, of course it’s our responsibility again to … share this information.”
The protocols for the tests, devised in 2009, were “thoroughly vetted by many dengue experts, including with WHO’s technical advisory group and various regulators”, Ng added.
The WHO said it acted promptly to address concerns when they arose. It has also said its recommendations on use of Dengvaxia were conditional and confined to children aged 9 and older in areas where dengue was endemic.
Complicating matters, scientists were sharply divided about the risks of the vaccine.
Four decades ago, Dr. Scott Halstead, a leading figure in dengue research, first proposed that antibodies from an initial exposure to one of four types of the disease could increase the risk of a potentially lethal complication called severe dengue when a person was infected a second time, a process know as antibody-dependent enhancement or ADE.
This phenomenon could make development of a dengue vaccine tricky.
VACCINE CAN ACT LIKE FIRST INFECTION
Rather than being protective, a shot given to someone who had never had dengue could act like a first infection, increasing their risk of severe dengue when they were exposed a second time.
Halstead, an adjunct professor at the Uniformed Services University of the Health Sciences in Bethesda, Maryland, said when he saw Sanofi’s 2015 paper, he was convinced the increased risk that some children would contract severe dengue was evidence of ADE.
Halstead wrote a series of papers published in Vaccine, the Journal of Infectious Diseases and other journals urging Sanofi and the WHO to proceed with caution in rolling out the vaccine. “It was not obvious to Sanofi and the World Health Organization, but it was obvious to me,” he told Reuters.
CONDITIONAL GUIDANCE
Dr. Joachim Hombach, senior health adviser in WHO’s vaccine department, said he understood Halstead’s concerns but the organization could only work with data generated by research.
He told Reuters that WHO “strongly encouraged” Sanofi to conduct follow-up studies to clarify whether children who had never had dengue before being vaccinated were more prone to severe dengue.
“It was recognized it was something that would be important to follow as part of our long-term program,” said Sanofi’s Ng. “This is exactly what we’ve done with our recent analysis.”
In the end, WHO issued its conditional guidance on Dengvaxia in July, 2016, after a year of deliberations, favoring use of the vaccine only in countries with a high burden of dengue, and stipulated that it only be given to children aged 9 and older.
Ferguson, at Imperial College, also raised concerns about the vaccine in a paper published in the journal Science in September 2016, suggesting the vaccine could increase severe dengue in some places with low transmission of dengue.
But until last month, many other scientists were on the fence about whether the signals in Sanofi’s trials were evidence of true ADE.
“I‘m not sure it was nailed down until now,” Jeremy Farrar, director of the Wellcome Trust medical charity who has served on WHO’s working group on dengue vaccines, told Reuters.
A paper in PLOS Medicine in November 2016 described a “split” among dengue experts between those like Farrar who supported the WHO recommendations on cautious use of Dengvaxia and those such as Halstead who said it was too risky.
To do the follow-up study, Sanofi had to solve a major problem. The company said it had only collected blood samples that would have revealed prior exposure to dengue in 10 percent of the more than 30,000 children in its clinical trials before they had been vaccinated.
Sanofi did have blood samples from nearly all of the children but only after they had been vaccinated. So it would be hard to tell from those samples whether the antibodies in their blood were produced in response to a natural dengue infection or the vaccine itself.
Responding to questions from Reuters, the company said in an email it would have been considered ‘unethical’ to take blood samples from all the kids prior to vaccination, because there wasn’t a clear reason to do so when it set up the protocols for the trial.
SHEER MOMENTUM
In 2014, Sanofi had entered into a scientific collaboration with the University of Pittsburgh Center for Vaccine Research to measure responses to the vaccine in its trials. At the time, the company had no reason to suspect that the vaccine would increase risks in children who had never had dengue, Ng said, but this test ultimately proved critical in allowing the company to differentiate those who had been infected before vaccination from those who had not.
That test only became ready to use this past July, said Dr. Ernesto Marques, an infectious disease specialist at the University of Pittsburgh, who helped develop the test.
Sanofi used the test to re-analyze blood samples collected in its trials and alerted public health officials last month that there was an increased risk of severe dengue in people who had not been previously infected, even among the children 9 and older who had been recommended for the vaccine.
By the time Sanofi announced findings from its follow-up study at the end of last month, the vaccine had been given to over 830,000 children in the Philippines, and about 300,000 people in Brazil.
One factor in decision-making was the sheer momentum behind the project, said Ferguson, the unpaid adviser, referring to the surge in the incidence of dengue and the fact that Dengvaxia was the first vaccine developed to contain the disease.
The WHO says dengue is now endemic in more than 100 countries, up from 9 countries before 1970. An estimated half a million people suffering from dengue require hospitalization each year and about 12,500 die.
“Clearly, in retrospect, it was a mistake” not to have taken baseline blood samples, said Ferguson, “The question is, could it (the need to do so) have been anticipated?”