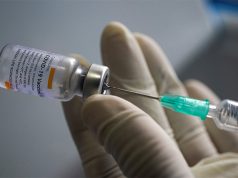
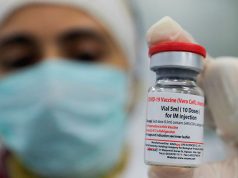
The Food and Drug Administration has approved the Pfizer-BioNTech‘s COVID-19 vaccine for emergency use days after the Palace said Filipinos who prefer to be inoculated with it will have to wait longer.
This was confirmed by FDA Director and Health Undersecretary Eric Domingo on Thursday.
The Pfizer-BioNTech was the first vaccine in the world that yielded a 95% success rate.
It was also the first one that the country’s FDA authorized for emergency use among the six other manufacturers that government officials are negotiating with, which including the vaccine from Chinese manufacturer Sinovac Biotech Ltd.
FDA explained that the emergency use authorization is “is an authorization issued for unregistered drugs and vaccines in a public health emergency” such as the COVID-19 pandemic.
The United States’ FDA added that under an EUA, the organization “may allow the use of unapproved medical products, or unapproved uses of approved medical products in an emergency to diagnose, treat, or prevent serious or life-threatening diseases or conditions.”
Following reports of the Pfizer vaccine approval, some Filipinos speculated who will be the first to receive the Pfizer jab.
Meanwhile, in a national address aired Wednesday evening, President Rodrigo Duterte defended his administration’s move to choose the Chinese-developed vaccines.
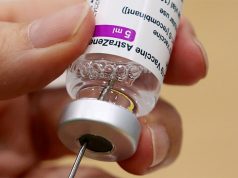
While the government had set its eyes on Sinovac, some local mayors and governors across the country have secured doses of AstraZeneca from the United Kingdom.
READ: Juxtaposed: National gov’t procures Sinovac vaccines while some LGUs ink deal with AstraZeneca
The Philippines has also inked deal for 30 million doses of Covovax from Serum Institute of India.
Aside from Pfizer, Sinovac, UK’s AstraZeneca and Russia’s Gamaleya have also applied for an EUA before the FDA.
On Sinovac
This development came after Palace defended the administration’s plan to purchase Sinovac vaccine or CoronaVac despite the lower efficacy rate and lack of FDA’s authorization.
The Philippines have already secured 25 million doses from the Beijing-based pharmaceutical firm and the initial batch is expected to arrive by February.
In a televised briefing on Wednesday night, January 13, Duterte claimed that Sinovac is as “good” as the rest of the vaccines being developed and that the Chinese pharmacists behind it are better.
“Now, the bakuna that Secretary Galvez is buying is as good as any other bakuna na naimbento ng mga Amerikano o mga Europeans. Hindi nagkulang ang Chinese, hindi sila nagkulang sa utak. Bright itong mga Intsik at they would not venture kung hindi sapat, it is not safe, sure, and secure. Iyong tatlo: it must be safe; sigurado, sure; and secure. That is the guarantee,” he said.
“The same microbes ang pinag-aralan nila so kanya-kanya ‘yan. It does not mean to say that the Americans or the Europeans, the EU, are better than the Chinese,” he added.
Duterte also made a confusing explanation on why the government should be the one choosing the COVID-19 vaccine brand.
“Ang gobyerno ang babagsak dito after sa bakuna ng iba, tapos may bumagsak na limang libong Pilipino, ililibing ako niyan. Not because namili sila ng pangit — hindi naman talaga…It is government who will say, ‘Sige, ito ang pinili mo, Brand A, titignan ko ha, sandali.’ Pagkatapos niyan, ‘Oh, okay ‘to,'” he said.
Early this week, presidential spokesperson Harry Roque likewise remarked that Filipinos should not be too picky with the vaccine brand, citing that it is administered to them for free.
“Wala pong pilian, wala po kasing pilitan… So tama lang naman po iyan ‘no, walang pilian kasi hindi naman natin maku-control talaga kung anong darating ‘no at libre po ito ‘no,” Roque said.
READ: ‘Filipinos deserve better’: Roque slammed for remark on choosing COVID-19 vaccine brand
Concerns on vaccine efficacy
In a Reuters report, experts cited that trial size, patient criteria, duration of post-vaccination observation, target groups and prevalence of the virus in the site are among the factors for the varying efficacy rates of vaccines.
So far, Pfizer-BioNtech yielded 95%, AstraZeneca has from 62% to 90%, Gamaleya has 91.4% and Moderna has 94% efficacy rates.
In the Philippines, Roque previously stated that facilities to store Pfizer’s vaccine are limited, thus only “major cities” in the country will gain access to it.
“Pati po sa Amerika na napakarami nilang na-order, nasasayang po dahil from the factory to delivery, hindi po nasusunod ang cold chain,” Roque said.