The vaccine type with inactivated virus like Sinovac’s CoronaVac prevented more hospitalizations and deaths, according to a new study.
The study “Effectiveness of an Inactivated SARS-CoV-2 Vaccine in Chile” from the New England Journal of Medicine was shared by vaccinologist Dr. Melvin Sanicas on September 9.
Sinovac is the national government’s preferred brand of COVID-19 vaccine and the first to arrive in the Philippines last February.
Chile, located in South America, is among the countries that also administered Sinovac’s CoronaVac to their residents.
“New! Sinovac’s CoronaVac inactivated COVIDvaccine study published in the @NEJM. The vaccine prevented hospitalizations by 87.5%, ICU admissions by 90.3%, death by 86.3% in >10.1 million people in Chile. This vaccine is saving lives! Let’s use it widely and save more lives!” Sanicas wrote.
“The results show that @Sinovac’s inactivated #SARSCoV2 #vaccine effectively prevented #Covid19, including severe disease & death – a finding that is consistent with results of phase 2 #clinicaltrial of the vaccine, he added.
‼️NEW‼️ #Sinovac‘s #CoronaVac inactivated #COVIDvaccine study published in the @NEJM. The #vaccine prevented hospitalizations by 87.5%, ICU admissions by 90.3%, death by 86.3% in >10.1 million people in #Chile. This vaccine is saving lives! Let’s use it widely & save more lives! pic.twitter.com/6gdzMsc4le
— Dr. Melvin Sanicas 🩺🔬 (@Vaccinologist) September 8, 2021
In the study, which link Sanicas provided on his tweet, the researchers stated that they used a “prospective national cohort” in their assessment that was conducted from February 2 to May 1, 2021.
“We used a prospective national cohort, including participants 16 years of age or older who were affiliated with the public national health care system, to assess the effectiveness of the inactivated SARS-CoV-2 vaccine with regard to preventing Covid-19 and related hospitalization, admission to the intensive care unit (ICU), and death,” they wrote.
It should be noted that the researchers did not specifically name Sinovac’s vaccine in their study.
Instead, they cited the type of COVID-19 jab— the inactivated SARS-CoV-2 vaccine.
Of the 10,187,720 participants, the researchers found that individuals who are fully-vaccinated received the most benefits of the COVID-19 shot.
They released a simple breakdown of the results in an infographic.
Based on this data, vaccine efficacy for fully-vaccinated participants are:
- Symptomatic COVID-19 – 65.9%
- Hospitalization – 87.5%
- Intensive care unit admission – 90.3%
- Death due to COVID-19 – 86.3%
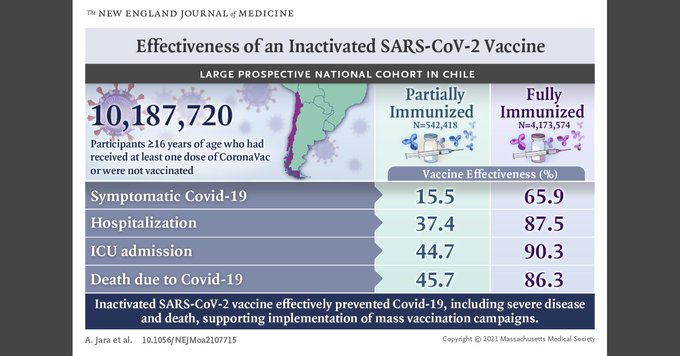
Based on the results, the researchers concluded that COVID-19 vaccines made from the inactivated virus are effective in preventing severe disease and death.
“Our results suggest that the inactivated SARS-CoV-2 vaccine effectively prevented Covid-19, including severe disease and death, a finding that is consistent with results of phase 2 trials of the vaccine,” they wrote.
Recap of the types of COVID-19 jabs
According to an infographic by the Boston University, there are seven basic types of vaccines. These are:
- Live attenuated vaccine – currently used in chickenpox
- Inactivated vaccine – currently used in polio, also by Sinovac, Sinopharm and Bharat
- Replicating viral vector vaccine – currently used in veterinary medicine
- Non-replicating viral vector vaccine – currently used in Ebola
- Subunit vaccine – Currently used in HPV, pertussis and hepatitis B
- DNA vaccine – Not currently known
- RNA vaccine – First used by Moderna, Pfizer-BioNTech, CureVac and Fosun Pharma
Last March, during the start of the country’s national inoculation program, Sanicas released an infographic of the seven manufacturers that developed vaccines to prevent COVID-19 and the corresponding vaccine types they made.
- Pfizer-BioNTech – mRNA in lipid nanoparticle, two doses
- Moderna – mRNA in lipid nanoparticle, two doses
- AstraZeneca-Oxford – Non-replicating chimp adenovirus, two doses
- Sinovac – inactivated SARS-CoV-2 coronavirus, two doses
- Gamaleya – Ad26 & Ad5 adenovirus vector, two doses
- Johnson&Johnson – Non-replicating human adenovirus, one dose
- Novavax – S protein/RBD; Martix M adjuvant, two dose
READ: Are these COVID-19 vaccines eyed by Philippines effective? A vaccinologist weighs in