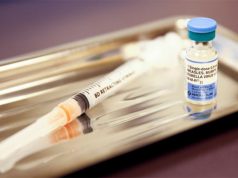
GENEVA/ZURICH — The world’s master list of recommended medicines got an update on Tuesday when the World Health Organization (WHO) published a biennial revision, adding 28 drugs including an abortion pill but leaving out several breakthrough treatments for lung cancer.
The WHO’s Essential Medicines List, which includes treatments that the WHO regards as global standards that should be available everywhere, aims to help governments make the best choices for their health systems.
The latest edition rejected Keytruda from Merck, Opdivo from Bristol-Myers Squibb and Tecentriq from Roche for non-small cell lung cancer, where they have combined for billions of dollars in sales in recent years.
“The Committee considered that their place in therapy for this condition is still evolving and that more data with longer follow-up are needed to better demonstrate estimates of their actual magnitude of benefit,” the WHO said.
It did recommend Keytruda and Opdivo, also known as pembrolizumab and nivolumab, for advanced melanoma, saying they had been shown to improve survival rates by 50% for a condition that was incurable until recently.
The WHO has a core list of 460 medicines, and a complementary list of treatments that require special care or facilities. It moved the abortion drug mifepristone off its complementary list and onto the core list, deleting a note that said it required “close medical supervision”.
However, it retained a note saying mifepristone should be used “where permitted under national law and where culturally acceptable”.
Nicola Magrini, the secretary of the WHO’s expert committee on selection and use of essential medicines, said there had been 65 applications for additions and about a third had been rejected.
Despite Tecentriq’s exclusion, Roche remains convinced of the merits of its so-called checkpoint inhibitor that helps take the brakes off the immune system, so it can attack cancer cells.
“We are disappointed that, on this occasion, our checkpoint inhibitor Tecentriq is not included in the EML,” the Basel-based drug maker said of the list.
“Complex treatments, such as those for cancer, often require sophisticated diagnosis, specialized training for healthcare professionals and hospital infrastructure for patients to get the care they need.”
The 2019 changes were based on health benefits available due to new research and new developments. Magrini said it normally took a few years for the recommendations to filter through into health systems.
Emerging biosimilars
Another Roche medicine, the $2.4 billion-per-year multiple sclerosis drug Ocrevus, also missed the cut along with rival MS treatments Gilenya, from Novartis, and glatiramer acetate, the generic name for Teva’s Copaxone.
The WHO concluded they did not clearly show superiority to other options, when considering factors including affordability. Ocrevus costs $65,000 annually, while Gilenya’s U.S. list price runs around $100,000.
Another rejected candidate was methylphenidate, branded as Ritalin, for attention-deficit hyperactivity disorder (ADHD), which Magrini said produced “unconvincing data on efficacy for long-term benefits”.
Among additions were anticoagulants for preventing stroke and treating deep vein thrombosis, giving an alternative to warfarin whose requirements for regular monitoring can be challenging in poorer countries.
Another was carbetocin for post-partum haemorrhage, an analogue for oxytocin but without oxytocin’s refrigeration requirements.
AbbVie’s $19-billion-per year blockbuster Humira, or adalimumab, as well as emerging copies, or biosimilars, of the world’s top selling medicine were added for rheumatoid arthritis and inflammatory disorders.
While adalimumab is costly, the committee concluded “increased availability of biosimilar products could lead to more market competition and reduced prices”.
The WHO list also included a vaccine for dengue fever for use in some high-risk populations.
The vaccine, Sanofi’s Dengvaxia, can increase the risk of severe dengue in children who were never exposed to the virus, and is at the center of a health scare in the Philippines, where the Department of Justice has said it has found probable cause to indict officials over 10 deaths.
Sanofi has repeatedly said Dengvaxia is safe and effective.
The WHO’s report on the list is available at this link.—Reporting by Tom Miles and John Miller in Zurich; Editing by Catherine Evans and Alison Williams